PGT is an umbrella term that includes different types of genetic evaluation. The specific type of testing recommended depends on the patient’s medical history and diagnostic findings.
PGT-A (Preimplantation Genetic Testing for Aneuploidy) evaluates embryos for abnormal numbers of chromosomes. Chromosomal abnormalities can affect embryo development and pregnancy outcomes.
PGT-A may be discussed in cases such as:
PGT-A does not guarantee implantation or pregnancy and does not test for all genetic conditions.


PGT-M is used when one or both partners are known carriers of a specific inherited genetic condition. This type of testing focuses on identifying embryos that do not carry the targeted condition.
PGT-M requires:
Because of its complexity, PGT-M is recommended only when there is a clear medical indication.
PGT-SR is used in cases where one partner carries chromosomal structural rearrangements, such as translocations. These rearrangements may increase the risk of miscarriage or implantation failure.
This form of testing helps identify embryos with balanced chromosomal structures but does not eliminate all reproductive risks.

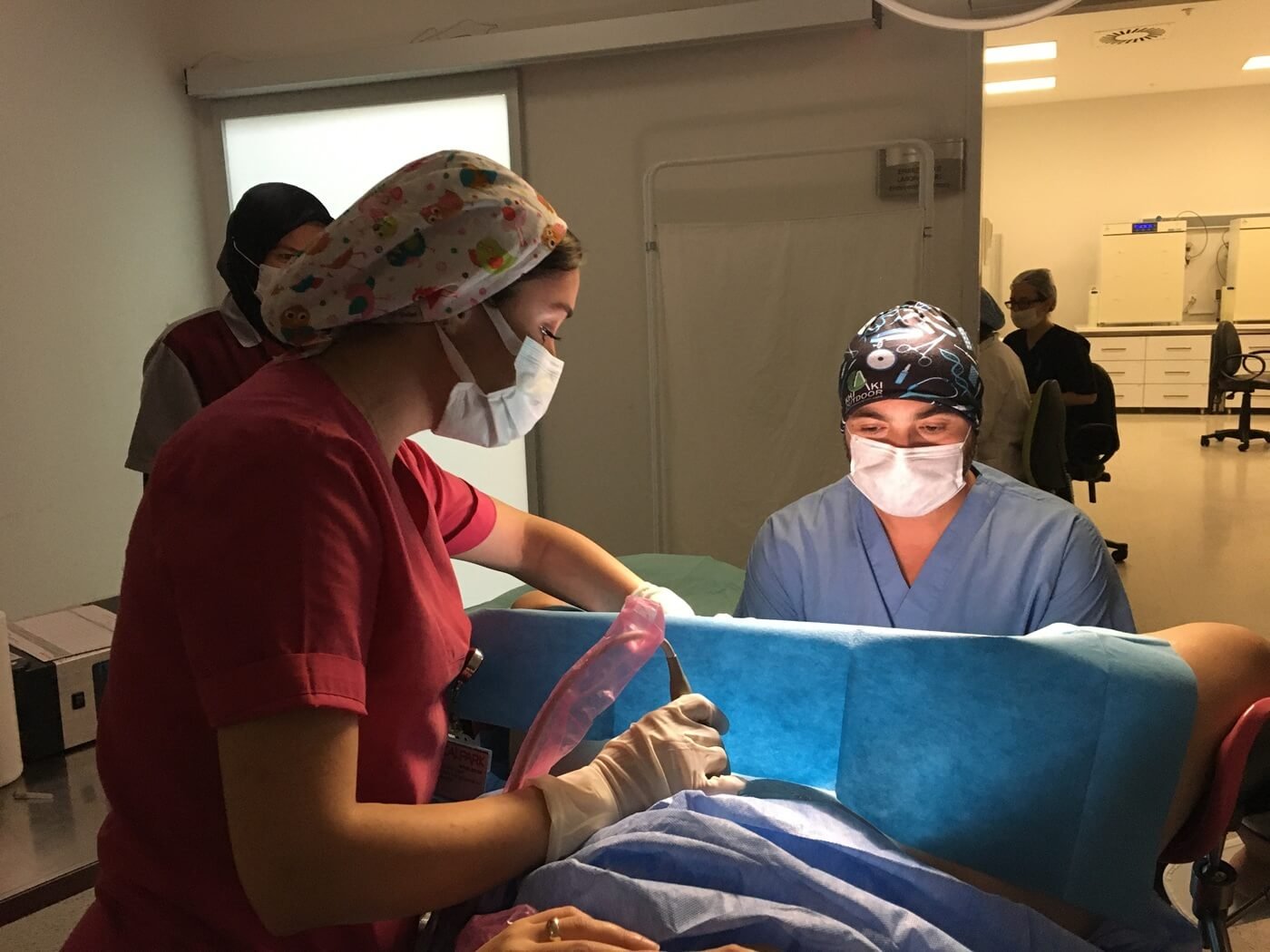
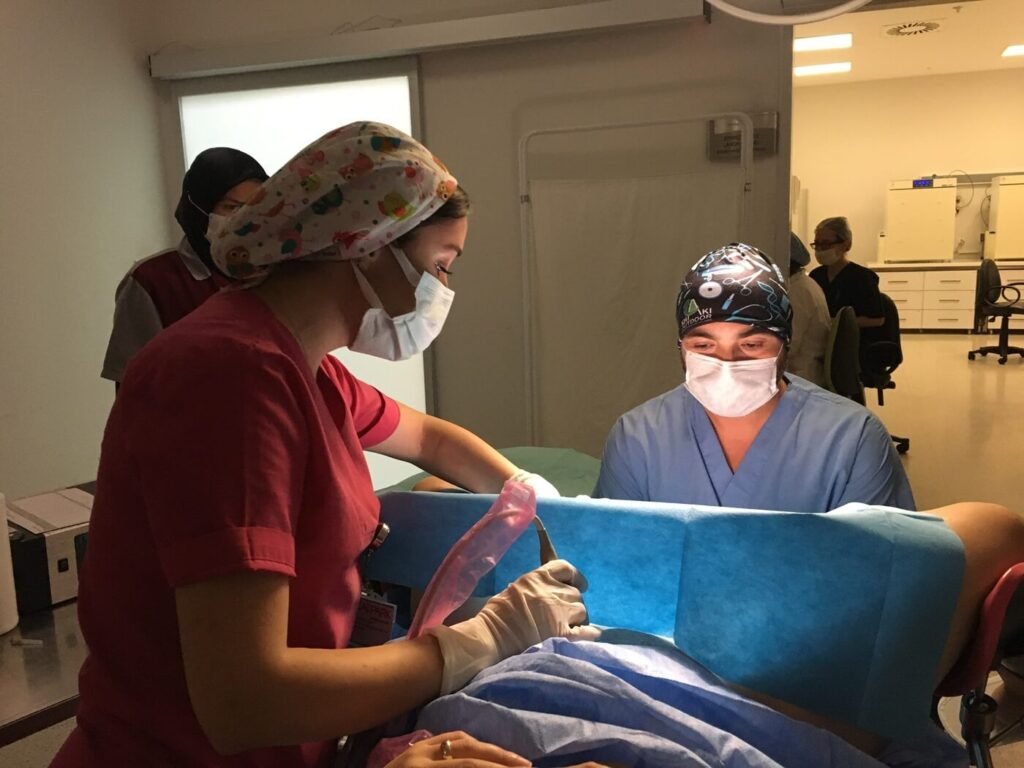
PGT is integrated into the IVF process and involves several coordinated steps within the laboratory.
PGT begins with a standard IVF cycle. Eggs are retrieved, fertilized in the laboratory, and embryos are allowed to develop over several days.
Embryo development is monitored closely by embryologists to determine suitability for biopsy.
At a specific stage of embryo development, a small number of cells are carefully removed for genetic analysis. The biopsy is performed using precise laboratory techniques designed to minimize impact on the embryo.
After biopsy, embryos are typically cryopreserved (frozen) while genetic testing is completed.
Biopsied cells are analyzed using specialized genetic testing methods. The analysis focuses on the specific genetic or chromosomal factors relevant to the patient’s situation.
Testing results are reviewed and discussed with the patient as part of treatment planning.
Based on genetic findings and overall embryo development, a plan is made for embryo transfer. Decisions are guided by medical evaluation and patient-specific factors rather than genetic results alone.
Not all embryos may be suitable for transfer, and outcomes cannot be guaranteed.


The cost of genetic testing varies depending on the type of PGT performed, laboratory complexity, and number of embryos tested.

Book your consultation with our fertility experts the first step toward your parenthood journey.